|
VAPOURS of an AlkAne are passed over a HOT CATALYST What is the chemical test to distinguish between AlkAne and AlkEne?īromine water stays ORANGE with an alkane but goes from Orange to COLOURLESS with an AlkEneĮxplain how fractional distillation worksĬrude oil is heated to 350Â☌ crude oil boil the VAPOURS cool as it rises up the tower VAPOURS condense at different levels according to the size of the molecules: the smaller molecules have LOWER boiling point and condense at TOP of towerĭescribe the cracking procedure (experiment) 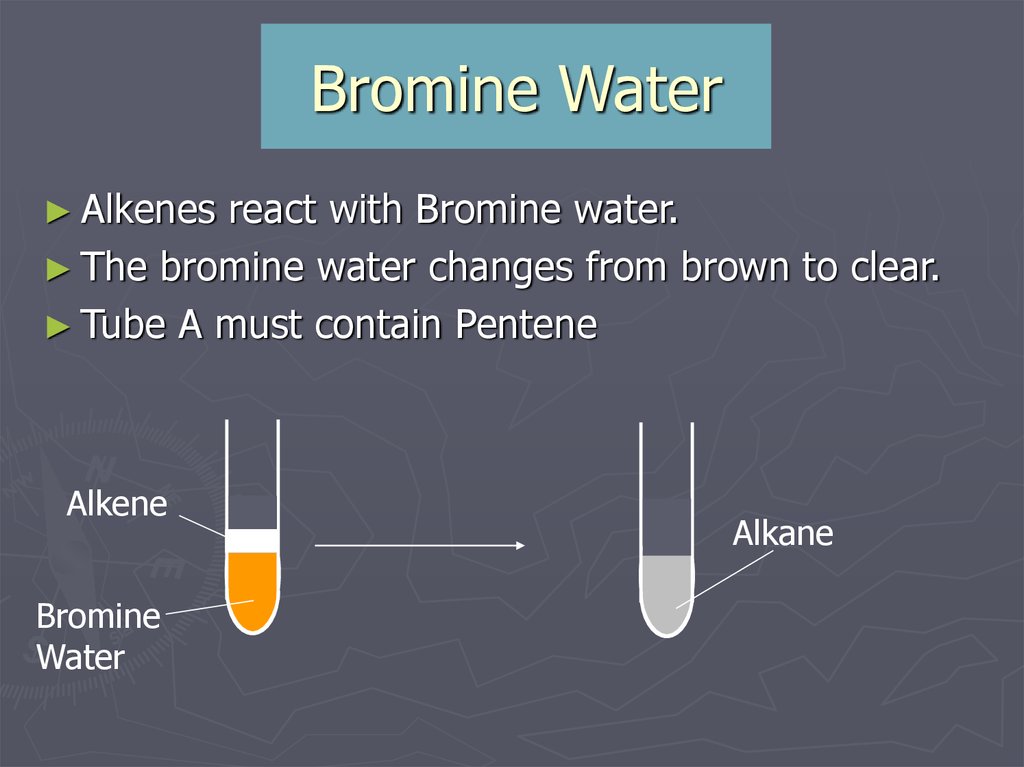
What is the mnemotechnic to remember that AlkEnes are Unsaturated?ĭEUX: Double Ene Unsaturated X (no more colour) What is the reaction where a long chain alkane breaks down into a shorter chain alkane and an Alkene?ĭraw the correct number of carbons in a chain join them with single links draw links so that each Carbon has FOUR links (no more, no less)ĭraw the correct number of carbons in a chain join TWO carbons with a DOUBLE link join the other carbons with single links draw links so that each Carbon has FOUR links (no more, no less) 
Which type of Hydrocarbons will make Bromine Water change colour? How long are the chains?Ĭh4 is the formula for a member of which family? The gases fraction contains chains of carbons. 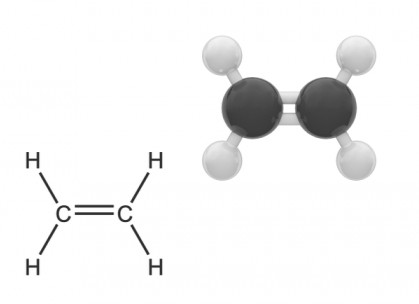
How do we separate the different fractions of crude oil? Which Hydrocarbons are the most flammable? long chains or short chains? GCSE5 crude oil AQA GCSE chapter 09 crude oil QuestionĪ COMPOUND of Hydrogen and Carbon ONLY, JOINED TOGETHER
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
